Advanced Assay Development LLC
Expert Consulting Solutions for
Development, Optimization & Validation
of Bioanalytical Assays or Platforms.
Immunoassay Development
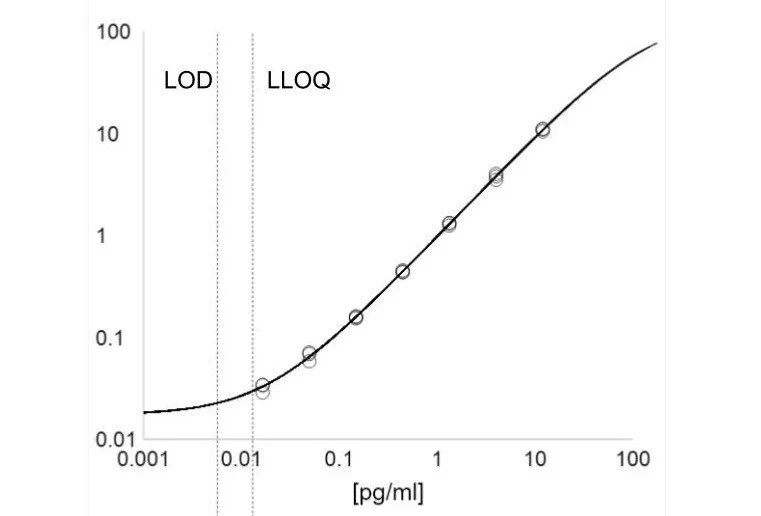
Immunoassays have been the workhorse of protein detection for over 60 years. I specialize in development, optimization and validation of basic ELISAs as well as leading technologies that enable better sensitivity (chemiluminescence, digital ELISA) and multiplexing (antibody arrays or proximity extension assays).
Specializing in
Antigen Selection
Antibody Screening
Multiplexing
Heterophilic Blockers
Buffer Optimization
Optimization of
Sensitivity & Specificity
Accuracy & Precision
Linearity & Dynamic Range
Quantitation
Cross-reactivity Minimization
Multiple Platforms including
Benchtop Elisa - Quanterix Simoa - Meso Scale Discovery ECL
Proximity Extenstion Assays (PEA) Olink & SPEAR - NuLISA (Alamar)
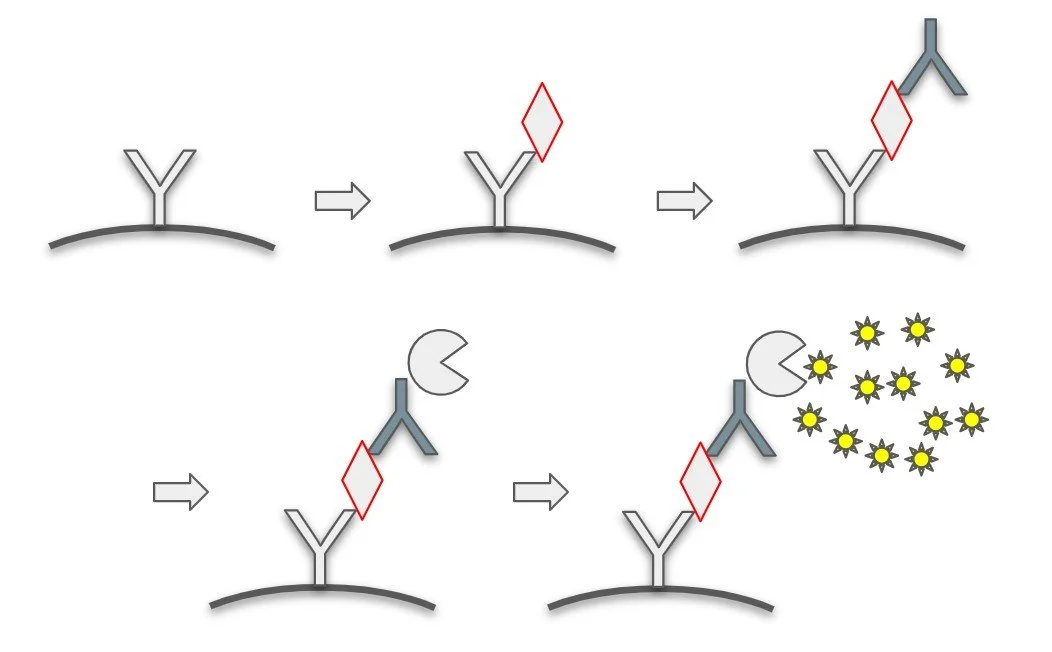
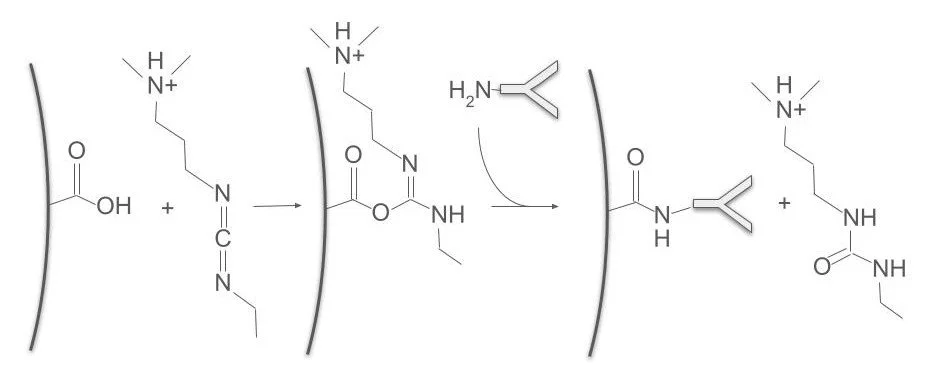
Bioconjugation and Bead Conjugation
Covalent labeling of proteins using a variety of chemistries… EDC/ S-NHS, click chemistry, carbohydate-modification etc.
Immobilization to bead and planar surfaces using covalent conjugation or adsorption. Passivation of surfaces to block non-specific binding. Surface modification to prevent bead aggregation, and monitoring of aggregation status.
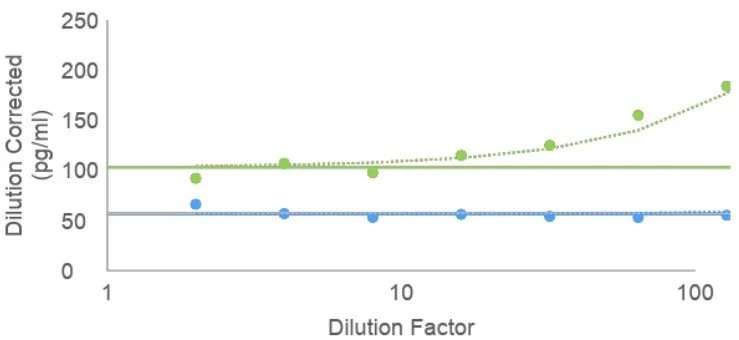
Assay Validation
Assays used for monitoring biomarker expression in clinical studies can be validated following Bioanalytical Method Validation for Biomarkers: Guidance for Industry (2025), whereas measurements of drug concentrations in bio-samples (PK/PD) should reference M10 Bioanalytical Method Validation and Study Sample Analysis: Guidance for Industry (2022). Immunogenicity testing follows Immunogenicity Testing of Therapeutic Protein Products — Developing and Validating Assays for Anti-Drug Antibody Detection (2019).
Following current guidance is crucial to ensure the correct tests are run to validate your assay. Adv Assay Dev can guide validation efforts from fit-for-purpose to CLIA to an FDA-approved Emergency Use Authorization.
Expertise in Extracellular Vesicles.
Extracellular vesicles (EVs) offer a promising platform for noninvasive diagnostics. All cells throughout the body release EVs into biofluids, packaging molecular cargo—including RNA and proteins—within lipid bilayers that preserve their integrity (Yu et al., 2021).
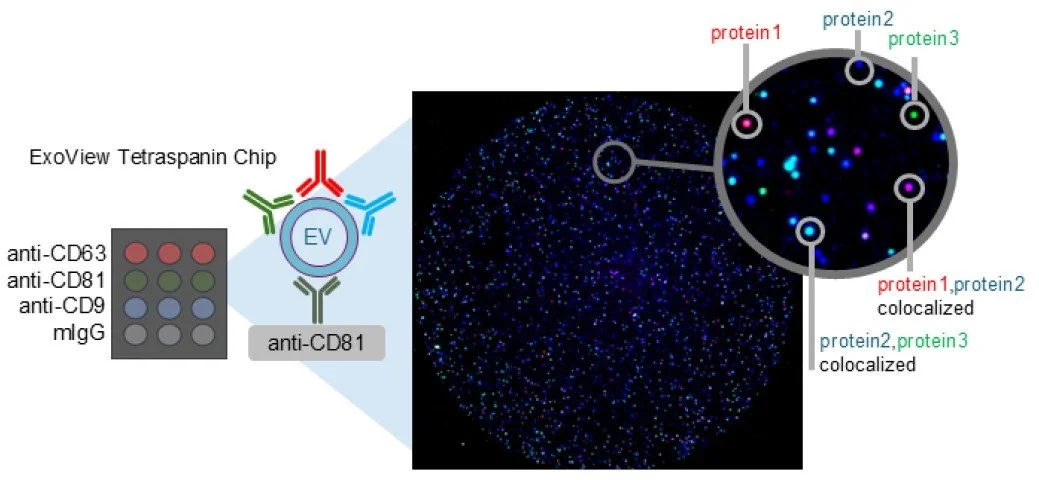
AdvancedAssayDev offers services for EV characterization, purification techniques and tissue-specific EV capture.
Platform Development
Development of novel platforms, such as automated immunoassays, ultrasensitive measurements, novel signal amplification technology.
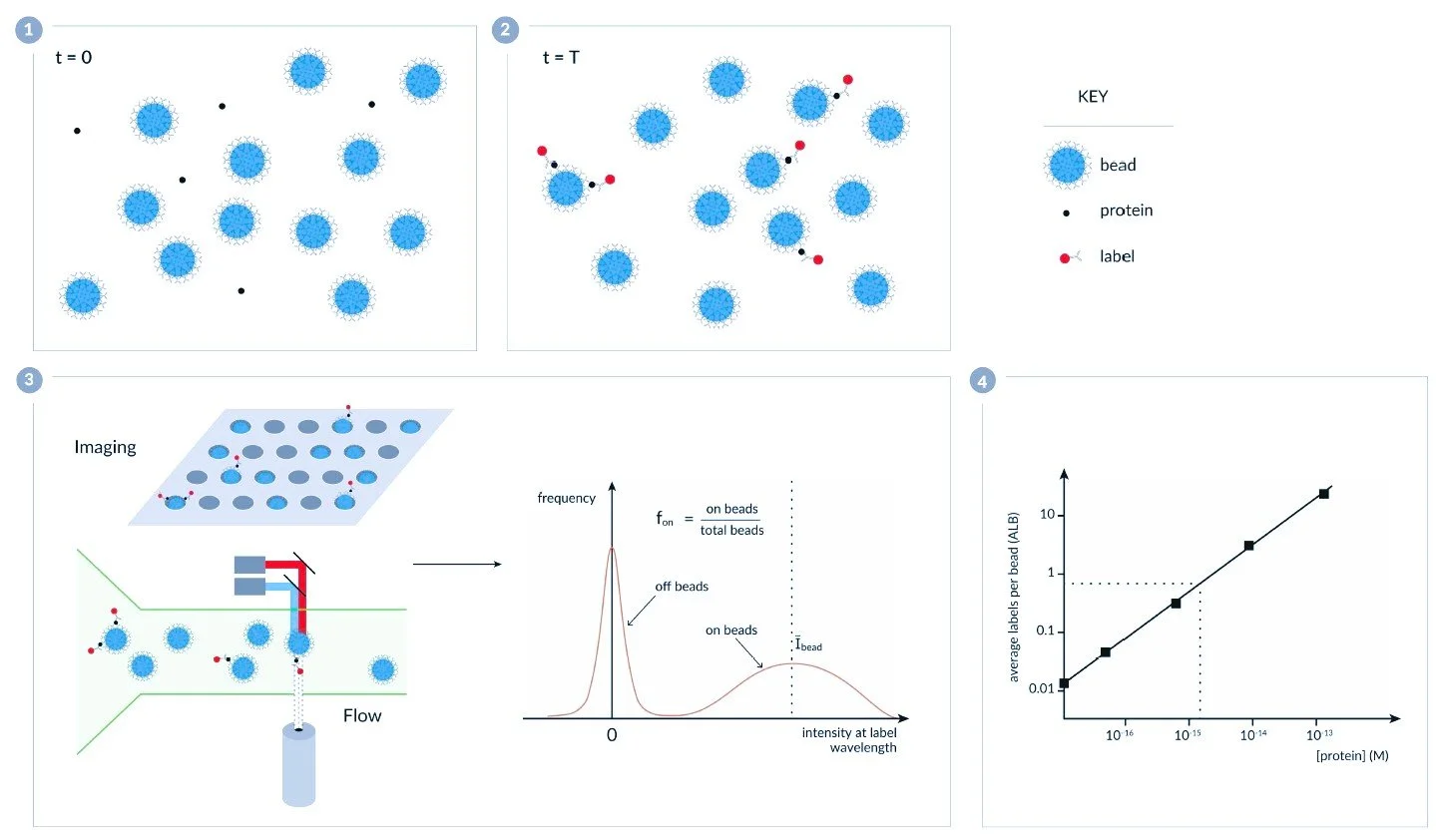
Proc. SPIE 12622, Optical Methods for Inspection, Characterization, and Imaging of Biomaterials VI, 126220I (11 August 2023); doi: 10.1117/12.2673470
PROFILE
With more than 14 years of experience in biotech, Joe started Advanced Assay Development LLC to help companies fulfill their mission of developing sensitive, specific ways to detect biomarkers of interest. He is an experienced scientist and technical leader in bioanalytical sciences, specializing in the development and implementation of fit-for-purpose assays.
Joe graduated from Stanford University with a PhD in biophysical chemistry. He has experience running biomarker labs in CRO and CLIA / CAP laboratories, as well assay and platform development of ultra-sensitive protein detection methodologies, extracellular vesicle detection and purification.

Contact Us
Interested in working together? Fill out some info and we will be in touch shortly. We can’t wait to hear from you!
